When life becomes an industry: jackpot or chaos?
- Synthetic biology is transforming our food supply, in particular through the creation of alternatives to animal proteins.
- This discipline combines biological and engineering knowledge, reshaping our relationship with health, the environment, energy and materials.
- DNA sequencing and editing, mathematical modelling and AI enable us to programme living organisms, balancing standardisation, modularity and decoupling.
- Impossible Foods uses yeast modified through synthetic biology to produce haem (which gives meat its taste and colour), to reduce the carbon footprint associated with livestock farming.
- The centralisation of digital data also applies to synthetic biology, where platforms such as Twist Bioscience play a key role in DNA synthesis.
The rise of alternatives to animal proteins, the proliferation of start-ups exploiting precision fermentation1. to produce ingredients identical to to their animal-based counterparts, and the growth of local biorefinery projects, all point to a tangible transformation within the biotechnology sector.
Resources allocated to innovation in fermentation and bio-preservation increased in 2024, with calls for proposals and public-private investments focusing on more efficient and sustainable industrial processes2. At the same time, specialist journals note that so-called “precision” fermentation techniques are revolutionising the production of food ingredients and establishing themselves as a driver of innovation capable of transforming the agri-food sector in the years to come3.
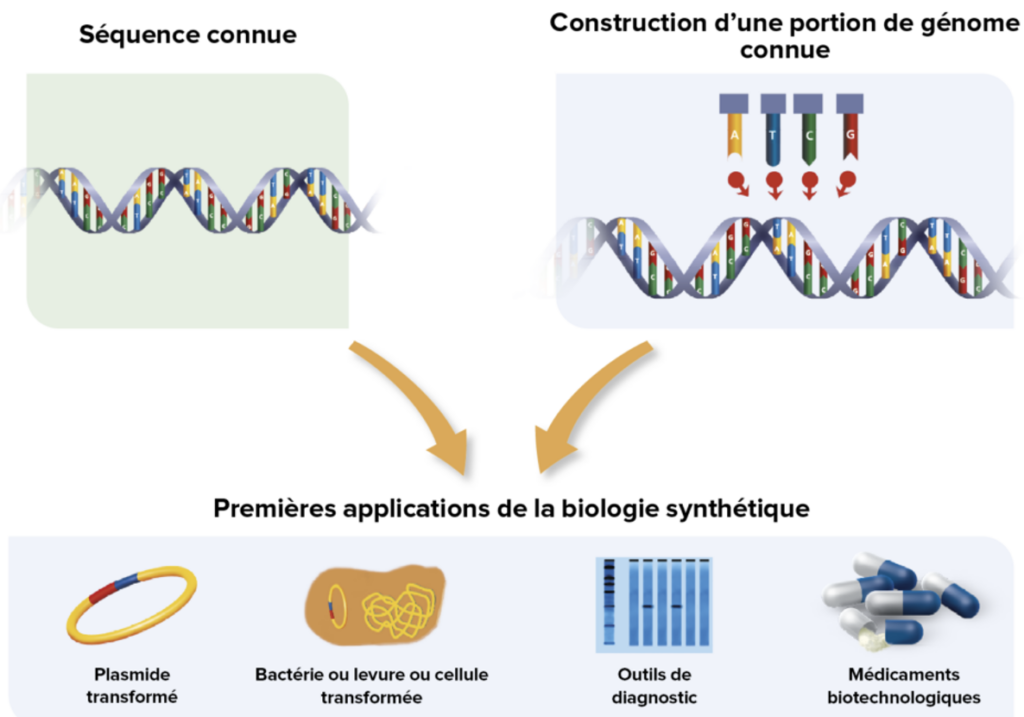
Emerging in the early 2000s, synthetic biology refers to the rational engineering of biological systems that goes beyond the incremental optimisation of traditional biotechnologies. It is based on principles of engineering, standardisation, modularity and the separation of design and manufacturing phases, made possible by advances such as high-throughput DNA sequencing, automated DNA synthesis, digital modelling, artificial intelligence and laboratory automation.
The aim is to construct organisms or biological modules that perform specific functions in a predictable manner, paving the way for new biomass-derived raw materials, bioreactor-based production units and value chains reorganised around living organisms rather than fossil resources4.
François Kepes has been at the heart of these developments for more than twenty years. A cell biologist and biotechnologist, and a member of the Académie des Technologies and the Académie d’Agriculture de France, he has helped to shape the field of systems and synthetic biology in France and Europe. His work focuses in particular on the modelling of complex biological systems and on synthetic biology. In 2011, he published the book “La Biologie de synthèse plus forte que la nature ?” (Is Synthetic Biology Stronger than Nature?), in which he analyses the scientific foundations, technological ambitions and industrial implications of this discipline. He has also authored several works on the scientific and economic dimensions of synthetic biology5, elucidating the conditions for its large-scale deployment.
#1 Synthetic biology represents a major breakthrough because it is based on engineering principles that enable the programming of living organisms, going beyond a purely incremental optimisation of existing biotechnologies
TRUE
Synthetic biology is the rational engineering of biology. The ambition of this field, which emerged in 2004, is to rationally design and construct, in a standardised manner, new systems inspired by biology or based on its components. Constructing a biological system that functions as intended is a way of ensuring that we have understood the underlying phenomena. In this sense, synthetic biology helps to advance our understanding of the living world. However, engineering plays a dominant role and touches on health, the environment, energy and materials.
Such a wide range of applications shows us that synthetic biology does not consist of a limited set of industrial solutions, but rather of a broad methodological and scientific foundation. This foundation is based on engineering principles that enable the “programming” of living organisms: standardisation, modularity and the decoupling of design and manufacturing. The conventional logic of incremental optimisation is giving way to rational design. The drivers of this paradigm shift in life sciences technologies are the large-scale adoption of DNA sequencing and editing, mathematical modelling and numerical simulation, artificial intelligence, and the automation of biology laboratories. As with the industrial revolutions of the past, beyond productivity gains, we are witnessing a paradigm shift: new raw materials (biomass), new factories (bioreactors), and new value chains.
The disruptive effect on production stems from the three points highlighted above: genericity, the programmability of living organisms, and the paradigm shift.
What does “programming life” actually mean?
Synthetic biology applies principles inspired by industrial engineering to living organisms:
- Standardisation of biological building blocks;
- Modularity of genetic functions;
- Separation between digital design and biological fabrication.
In practical terms, an organism becomes a production platform designed through computer modelling, whose DNA is synthesised and then assembled in a robotised laboratory. This model brings biological production closer to a “design – build – test – learn cycle6”, similar to digital industrial processes.
Innovation no longer focuses solely on raw materials, but on the ability to design optimised biological architectures before they go into production.
#2 The ability to programme living organisms tends to shift value creation towards the design phase of producing strains, upstream of processing chains
TRUE
Programming living organisms shifts value creation upstream in value chains: the design of production organisms becomes the key to competitiveness, rather than the processing of materials. For example, in the agri-food sector, companies such as Impossible Foods use modified yeasts, applying the principles of synthetic biology to produce haem (the molecule that gives meat its colour and taste). This significantly reduces the environmental footprint associated with animal husbandry. This shift in the point of value creation is accompanied by vertical integration.
To capture a large share of the value, companies must master the design of producing strains, industrial-scale fermentation and market launch. This is why the giants of the chemical and energy sectors are investing heavily in these technologies to avoid being left behind. Nevertheless, this transition also opens the door to local initiatives, centred, for example, on local biorefineries using microorganisms to recover waste produced in a given region.
What economic mechanisms shape synthetic biology?
Synthetic biology is reshaping value creation through three key dynamics:
Shift upstream: The design of producing strains is becoming the primary driver of competitiveness. Comparative advantage now lies in biological design, algorithms, modelling and intellectual property, rather than in the physical processing of raw materials.
Platform effects: Certain companies are structuring technological ecosystems that centralise genetic data, DNA synthesis capabilities and robotic infrastructure. These platforms can generate economies of scale comparable to those seen in the digital sector, without, however, freeing themselves from material constraints.
Industrial hybridisation: Unlike purely digital industries, biological production remains rooted in heavy physical infrastructure. The value chain therefore combines strategic intangible assets with localised industrial capabilities, creating hybrid models that balance global concentration with local roots.
This combination distinguishes the life sciences economy from previous technological waves and explains the industrial sovereignty issues discussed below.
#3 The business model of synthetic biology fully replicates the concentration of value seen in the digital sector, regardless of physical constraints
FALSE
Despite being relatively new, some synthetic biology platforms are reminiscent of the tech giants. Indeed, they centralise vast amounts of data and impressive equipment. For example, Twist Bioscience has rapidly established itself as a global leader in the field of DNA synthesis. With a very different business model, Ginkgo Bioworks offers a “cell factory” where, without their own infrastructure, its clients can design and manufacture bespoke strains of microorganisms. This concentration in large platforms is explained by the high costs of R&D and the need for access to rapidly growing databases.
However, unlike computational processes, those in synthetic biology rely heavily on physical resources. Indeed, the production of biomolecules requires logistics, floor space, fermenters and carbon sources typically derived from biomass. This allows for hybrid models, where global platforms coexist with local players specialising in production or adaptation to niche markets.
#4 Does Europe currently have full control over the tangible and intangible strategic assets associated with the ‘industrialisation of life?’
UNCLEAR
The ‘industrialisation of life’ shifts the focus of sovereignty to two types of assets: tangible (e.g. robotic platforms and massive bioreactors, strain collections and biobanks) and intangible (patented genetic sequences, biological databases, design algorithms). The governance of intangible assets raises issues of intellectual property, security and sharing. That of tangible assets raises industrial challenges on a par with those of batteries. Value capture occurs where the production capacity lies, which is not necessarily where high-quality research is conducted.
Two categories of assets underpin the new life sciences economy:
Tangible assets:
- Robotic platforms;
- Industrial bioreactors;
- Biobanks and strain collections;
- Large-scale fermentation infrastructure.
Intangible assets:
- Patented genetic sequences;
- Biological databases;
- Design algorithms;
- Simulation models.
Value capture depends on the combined mastery of these two dimensions. Research alone is not enough if industrial capabilities are located elsewhere.
#5 The environmental promises of synthetic biology are already economically viable on a large scale
FALSE
From a scientific perspective, pilot projects generally demonstrate the relevance and feasibility of their objectives, whether these involve, for example, the recovery of agricultural waste, carbon dioxide sequestration or the production of bio-based molecules. From an economic perspective, the entire life cycle of the processes must be taken into account. The critical parameters are energy consumption, the reliable availability of biomass, yields of the final product, and the genetic stability of the producing strains.
The environmental footprint of synthetic biology as such has not been the subject of in-depth studies, although it is generally accepted that its waste is compostable and that the water consumed is recovered at the end of the cycle. In addition to these technological barriers, there are external issues such as regulation, social acceptability, and carbon and energy prices.